It's also used in the production of batteries. 
It has a wide range of applications in manufacturing since it is the hardest substance known to man.Īmorphous carbon is used in inks and paints because it is a softer type of carbon. However, diamonds are used in the industry for uses other than jewellery. In jewellery, the diamond, which is the hardest form of carbon, is used. Carbon atoms make up carbohydrate molecules. The following are some of the most important applications:Ĭarbohydrates are a major source of energy in the foods we consume. The carbon cycle is a biological process that involves the conversion of atmospheric carbon dioxide to carbohydrates in plants, the consumption of these carbohydrates by animals and their oxidation through metabolism to produce carbon dioxide and other products, and the release of carbon dioxide back into the atmosphere.Ĭarbon, whether elemental or mixed, is usually quantified by turning it to carbon dioxide gas, which can then be absorbed by other chemicals to generate a titratable acidic solution or a weighable product.Ĭarbon is employed in so many daily tasks that one may not be able to even realise the true extent of it. Carbon is plentiful in coal, as well as the organic compounds that make up petroleum, natural gas, and all plant and animal tissue. The major component of coral and the shells of oysters and clams is calcium carbonate. This permits carbon to form lengthy chains and rings of atoms, which serve as the structural foundation for many of the molecules that make up a living cell, including DNA, which is the most important of them all.Ĭommon minerals (e.g., magnesium and calcium carbonates) are made up of carbon compounds (e.g., magnesite, dolomite, marble, or limestone). At the very least, we know about a million organic components, and this number is continuously increasing every year.Ĭarbon is also the most essential element in the universe since it is the only element capable of forming strong single bonds with itself that are chemically resistant under normal conditions. The greatest of these components is the group formed by carbon and hydrogen. Diamond is the most valuable and beautiful of all-natural gemstones, as well as the hardest of all naturally occurring abrasives.Ĭarbon's chemical characteristics are unique in that it produces a number of components that are superior to the total of all other elements. On the other hand, graphite which is also made up of carbon is a soft, slick solid that is a good heat and electrical conductor. The hardest naturally occurring solid is a pure diamond, which is also a poor conductor of electricity. Other compounds, such as carbon black, charcoal, lampblack, coal, and coke, are sometimes referred to as amorphous, however, X-ray analysis has indicated that they have a low degree of crystallinity.ĭiamond and graphite are found naturally on Earth and can also be synthesized they are chemically inert but, like amorphous carbon, mix with oxygen at high temperatures. 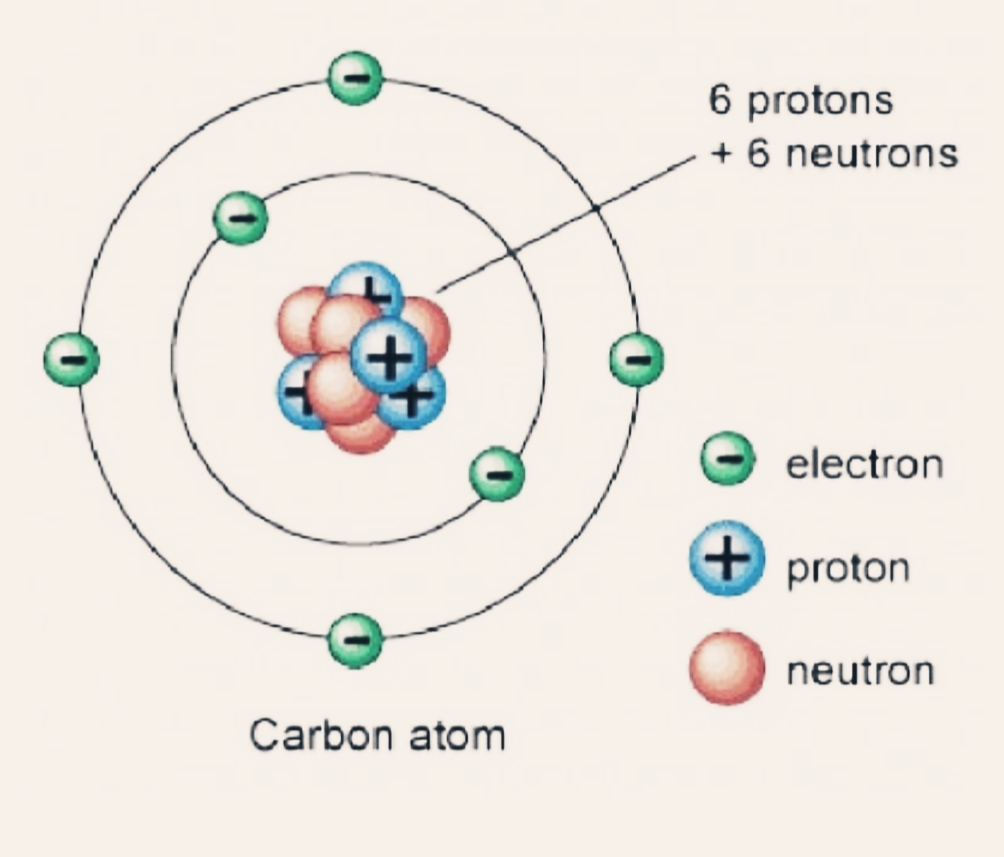
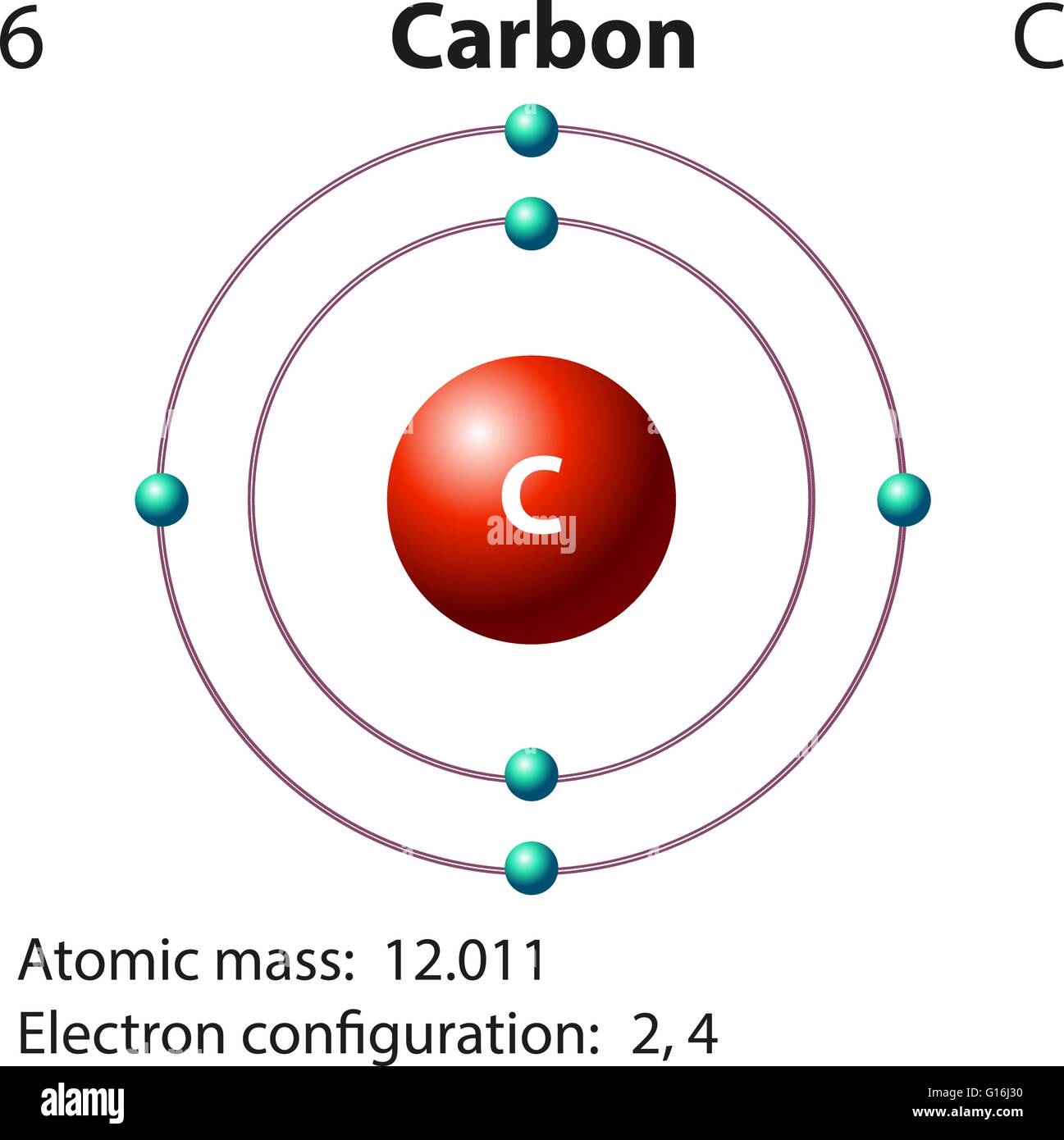
Fullerene, a third kind, is made up of a range of carbon-based molecules.Īmorphous carbon, on the other hand, has no crystalline structure. Although diamond and graphite, two of their well-defined forms, have crystalline structures, their physical properties differ due to the varied arrangements of atoms in their structures. Properties of Carbon Physical Properties of CarbonĬarbon is one of the most plentiful elements in the Earth's crust by weight.Ĭarbon is the cosmic product of helium's " burning ," in which three four-atomic-weight helium nuclei fuse to generate a twelve-atomic-weight carbon nucleus.Įlemental carbon is available in a number of different forms, each with its own set of physical qualities. The stable isotopes 12C and 13C, as well as the radioactive 14C, make up nature's isotopes. 
Carbon makes up only about 0.025 percent of the Earth's crust. It belongs to the modern periodic table's 14th group. It's nonmetallic and tetravalent, which means it can bond chemically with four electrons. Let us understand more about carbon atoms, carbon uses, and carbon compound information in everyday life.Ĭarbon is a chemical element with the atomic number 6 and the symbol C. Not only that, but carbon is the most abundant element in all organic matter. Carbon is the most important component of this chemical. Do you know what that is? Plastic may be found in everything from computer, smartphone, and tablet screens to clothing, food, and automobiles. Everything in your environment is made up of or contains one element in common. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
